No
Readiness to Receive Electronic Case Reports
As of November 1, 2021, the Nebraska Department of Health and Human Services (DHHS) declared readiness to begin receiving electronic case reports (eCR) for all reportable diseases.
Who is eligible to participate?
Eligible providers, critical access hospitals, hospitals.
What is eCR?
Electronic case reporting (eCR) is the automatic real time electronic data sharing of case report information between Electronic Health Records (EHRs) and public health authorities for disease tracking. It moves critical clinical and demographic data quickly and securely and helps eliminate the need for manual provider reporting. eCR offers several advantages, including reduced administrative burden of reporting and improved timeliness, accuracy, and completeness of data. This real time exchange of information is critical during public health emergencies.
How eCR works
The patient is diagnosed with a reportable condition such as COVID-19. The healthcare provider enters the patient's information into their EHR. The eCR triggers the transfer of clinical data from the EHR to a shared services platform called AIMS Hub. This occurs behind the scenes, without disrupting the healthcare providers' workflow. The message, known as the electronic initial case report (eICR), is then evaluated by a centralized decision support service to determine whether the potential case meets criteria for reporting; refer to the Reportable Condition Knowledge Management System link below for more information. If a condition is found to be reportable, the appropriate public health jurisdiction(s) receives the eICR in real time. In addition, a Reportability Response (RR) is sent back to the reporter to communicate information on the status of the reportable condition(s) and other relevant information. This might include additional follow up steps such as the need to collect specimens for testing at public health laboratories.
Initial Case Report Standards
Nebraska uses the HL7 electronic initial case report (eICR) standards (R1.1 and R3.1) for electronic case reporting (eCR) and to support the new CMS Promoting Interoperability requirements for eCR. Nebraska requires the use of APHL AIMS and the Reportable Condition Knowledge Management System (RCKMS). Use of these standards will eventually eliminate manual reporting requirements.
Getting Started
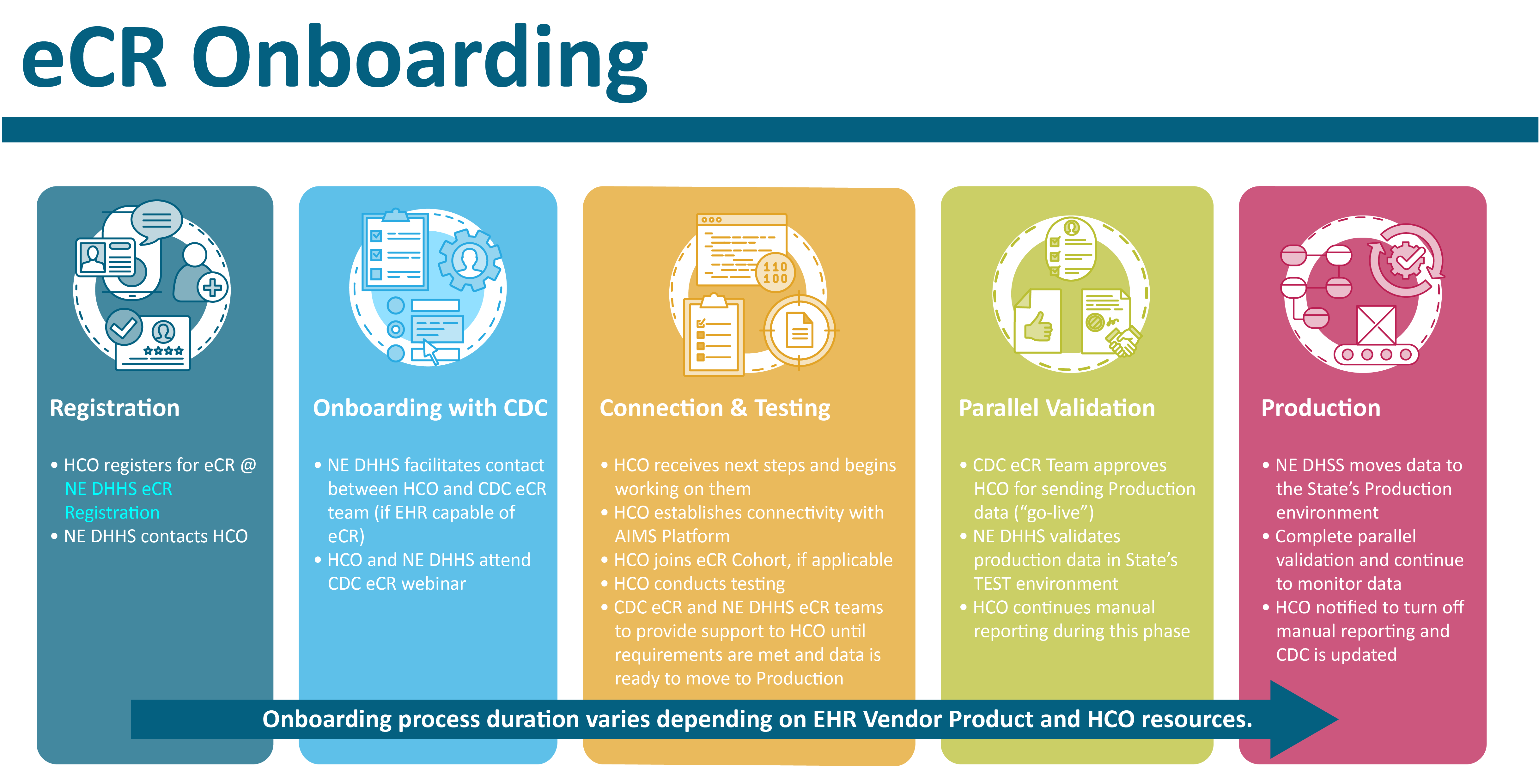 View the accessible PDF version.
View the accessible PDF version.
Register Here
- Complete the registration form.
- Refer to this web site https://ecr.aimsplatform.org/healthcare/ for additional information.
- Contact dhhs.ecr@nebraska.gov for more information.
Additional Resources
eCR is a collaborative effort between the Association of Public Health Laboratories (APHL), the Centers for Disease Control and Prevention (CDC) and the Council for State and Territorial Epidemiologists (CSTE).
For more information, visit: